How can one decision deliver improved patient safety, higher job satisfaction of lab staff, and save over 3,500 labor hours?

Background
A frequent misconception in the histology/pathology space is that only reference labs can benefit from investments in automation and advanced technology. However this is untrue as evidenced by the adoption of the Fina and Crystal systems at a leading academic medical center (AMC) in Boston, Massachusetts.
This AMC was established in 1996, but has roots all the way back to 1864. They are the largest safety net hospital in New England and a Medicaid Managed Care Organization. Also being the largest trauma center in New England, this is a bustling community of patient-centered experts. As a leader in clinical treatment and research, they received >$82 million in research funding from NIH. They have 789 ongoing research projects, including both laboratory based research and clinical research. Their daily volumes are between 90 – 100 surgical cases, 350 – 450 tissue blocks, and 750 – 900 slides.
Challenges
- Increasing case complexity with a greater number of blocks per case. This is a byproduct of delayed screening from the COVID pandemic. As patients catch-up to their cancer screenings, the delay from 2020 forward is contributing to more diagnoses of advanced disease. Consequently, there has been an increase in clinical trial participation and increased demand for sample retrieval to qualify for trials.

«With the increasing rate of clinical
Zahra Aryan. Asset Manager
trial participation, the worst call you can make in the Asset Management Services is telling a patient that you do not have their block and they cannot enter a trial.»
2. The scarcity of qualified HT/HTL labor has been noted all over the world. While this AMC is currently fully staffed, there is concern around staff retention, and attracting new staff as they experience caseload growth. Key to retaining and attracting new staff is creating a culture where lab staff are supported and equipped to do valuable work. This AMC wanted to stop having their lab staff perform mundane and manual activities that have low impact on patient care.
Decision
After extensive investigation of solutions to these challenges, in Summer 2023 they adopted two new systems from Dreampath Diagnostics:
- Fina system (for tissue block management)
- Crystal system (for microscope slide management)
Results
Implementation of the Fina system saved the lab over 1,500 hours annually in tissue block management.
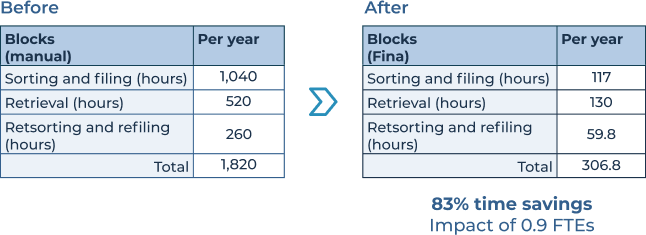
Implementation of the Fina system saved the lab over 1,500 hours annually in tissue block management.
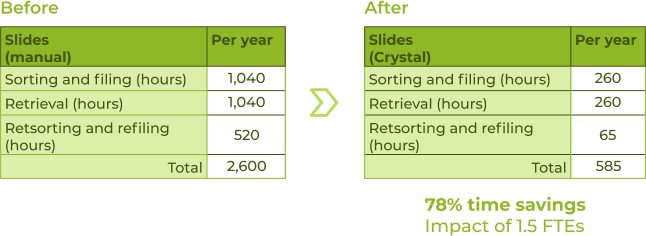
Beyond the time savings and confidence gained in proper sample tracking, archival and retrieval, the lab experienced several additional benefits:
- Lean workflow improvements through fewer touch points and wait points
- Error reduction by eliminating manual touch points
- Preparation for LIS transition to Epic Beaker (dictionary audits and current review of upfront protocols)
- Improved organization of samples that are part of pathologist research projects
- Ability to query trends and run reports on blocks not returned within one month
- Improved lab staff job satisfaction through eliminating mundane tasks. Higher focus on quality of work



